How to Read a Blood Panel Report
The U.S. Department of Wellness and Human Services (HHS) issued a final rule in 2014 that allows patients or their representatives direct access to laboratory test reports after having their identities verified, without the need to have the tests sent to a health practitioner get-go. This rule is intended to empower you, to allow y'all to human action as a partner with your healthcare provider and have a more than agile role in your healthcare decisions.
Easier access to exam results, however, places you in a position of greater responsibleness. You lot may see complex test results on lab reports and will need to recognize that there is a context in which providers use results to brand treatment decisions. This may require that you lot brainwash yourself about your tests in order to sympathize their purpose and meaning. Testing.com and other credible sources of health information online can aid you lot in achieving a amend agreement of your medical data.
Patients who want to tin can all the same get their exam results from their wellness practitioners and patients should still look to them equally the ultimate informed partner for understanding test results and providing handling options. Testing.com encourages you to talk over your lab test results with your health practitioner, using this web site to assist formulate your questions.
Once yous receive or access your written report from the laboratory, information technology may not be piece of cake for yous to read or understand, leaving you lot with more than questions than answers. This article points out some of the unlike sections that may be found on a typical lab report and explains some of the information that may be plant in those sections.
Sample Lab Reports
Dissimilar laboratories generate reports that tin vary greatly in appearance and in the society and kind of information included. Below are some examples of what lab reports may wait like. Select a sample study beneath to view or download information technology.
(Notation: Pathology reports, such as for abiopsy, will look different than this sample lab study. The College of American Pathologists provides a resource: How to Read Your Pathology Study.)
Select to View or Download Sample Reports
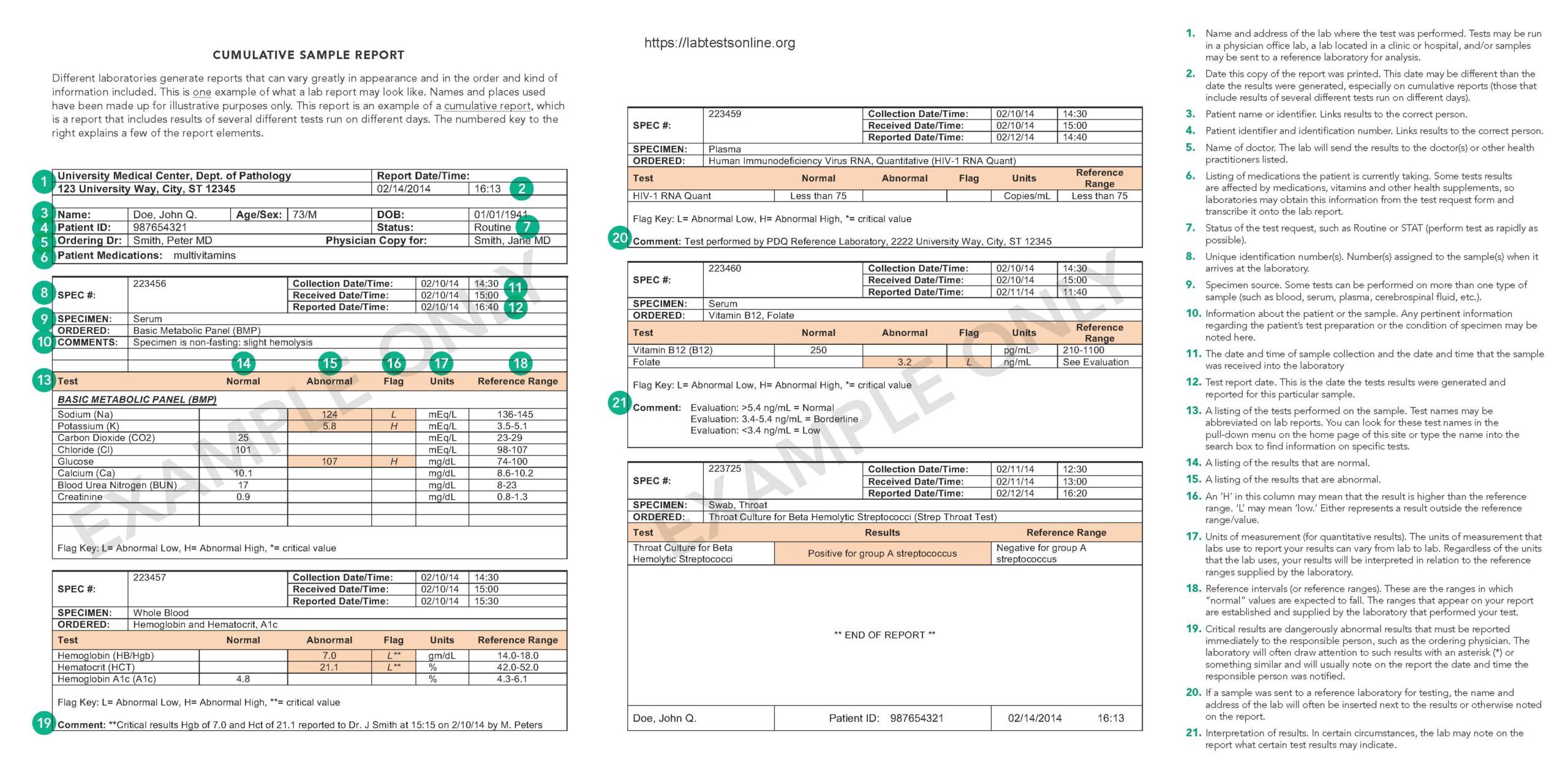
Sample Cumulative Study
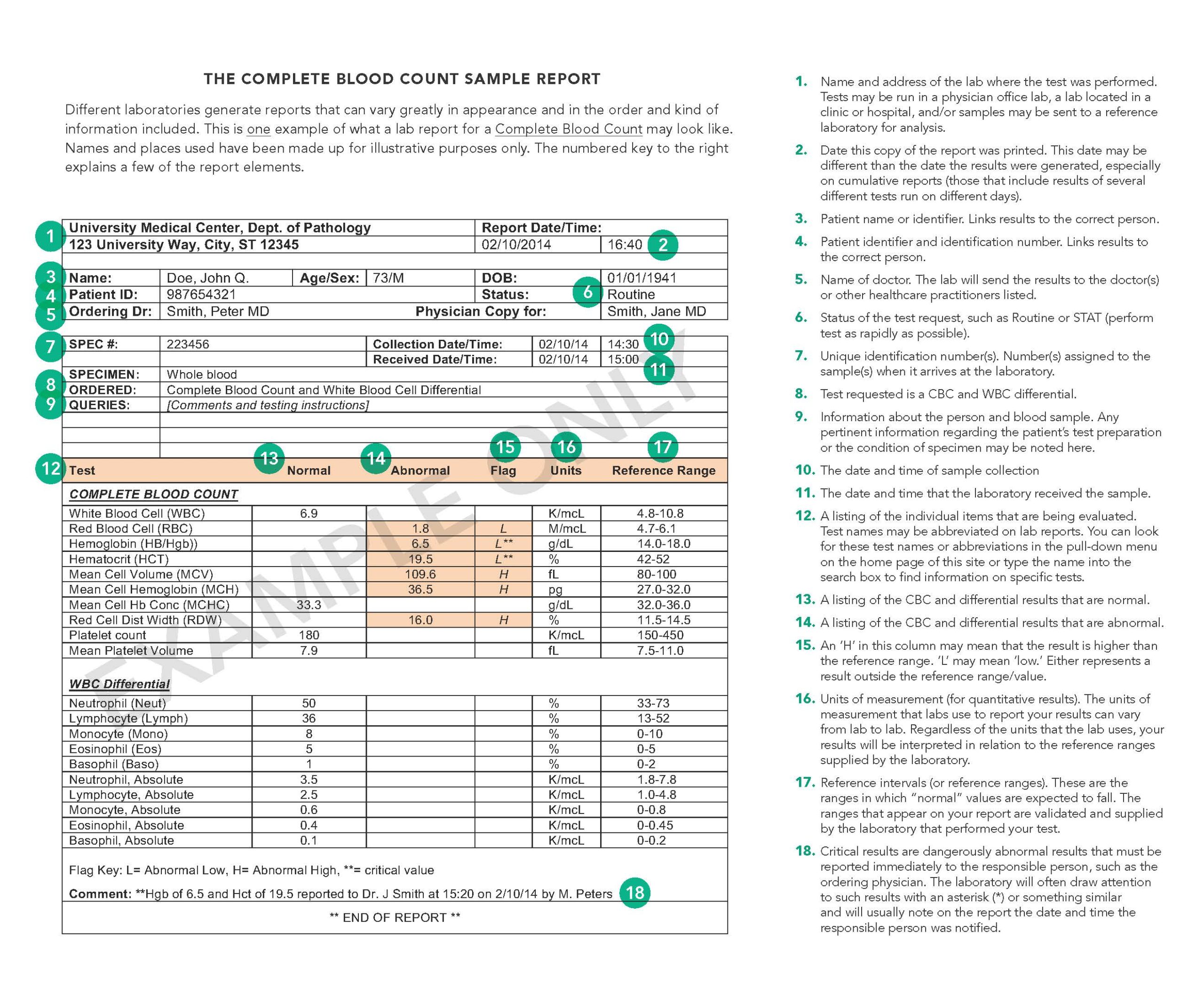
Sample CBC Written report
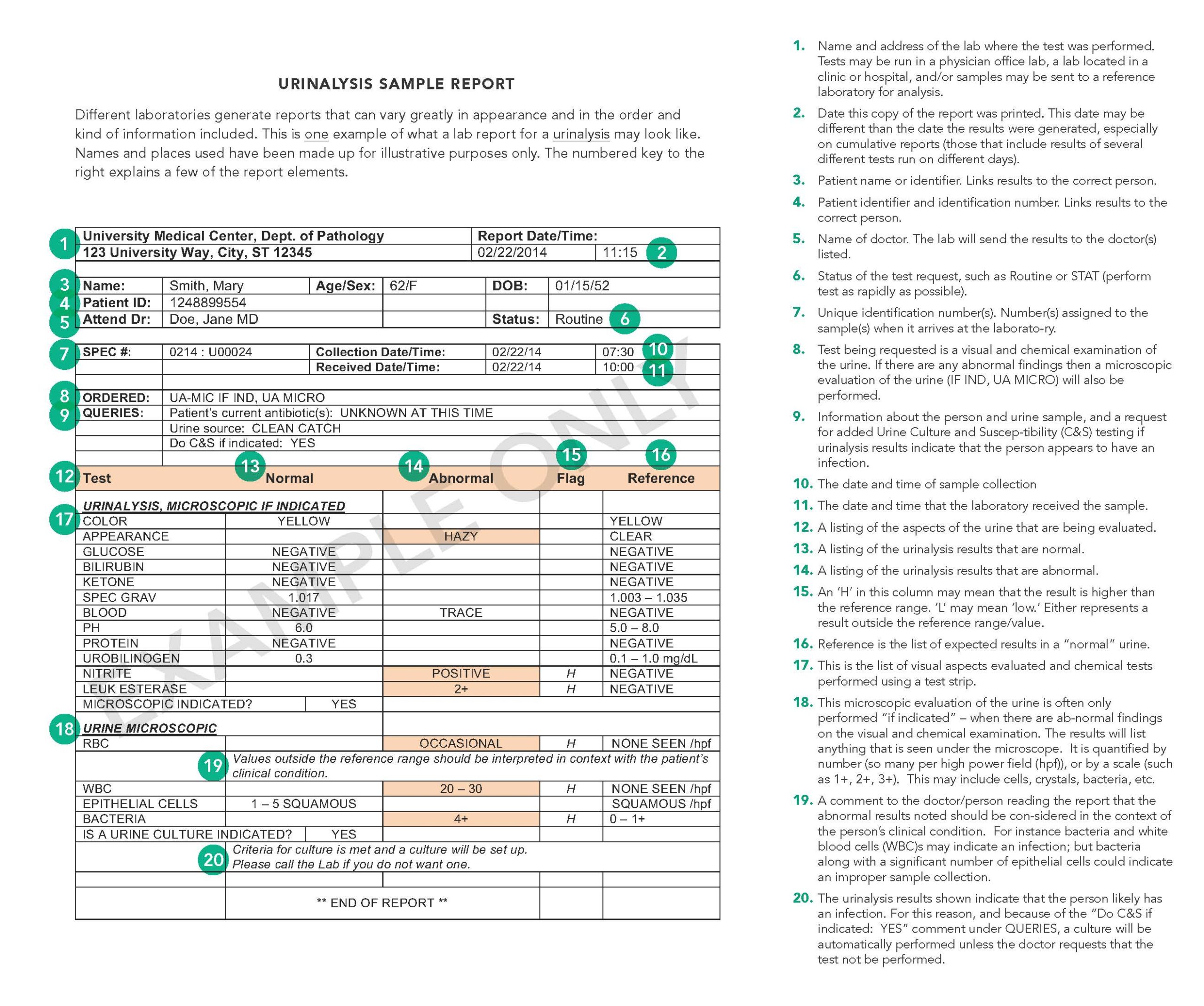
Sample Urinalysis Report
Components of a Typical Lab Report
Despite the differences in format and presentation, all laboratory reports must contain certain elements as mandated past federal legislation known as the Clinical Laboratory Comeback Amendments (CLIA). (CLIA '88 REGULATIONS, Department 493.1291; for more on regulation of laboratories, see Lab Oversight: A Building Cake of Trust.) Your lab study may look very different than the sample written report, simply it will contain each of the elements required by CLIA. It may as well contain additional items not specifically required just which the lab chooses to include to assistance in the timely reporting, delivery, and interpretation of your results.
Some items included on lab reports bargain with administrative or clerical information:
- Patient proper name and identification number or a unique patient identifier and identification number. These are required for proper patient identification and to ensure that the test results included in the report are correctly linked to the patient on whom the tests were run.
- Name and accost of the laboratory location where the exam was performed. Tests may exist run in a physician office laboratory, a laboratory located in a clinic or hospital, and/or samples may be sent to a reference laboratory for analysis.
- Date written report printed. This is the date this copy of the report was printed. Often, the time that the written report was printed will as well be included. The date of printing may be different than the appointment the results were generated (see beneath), especially on cumulative reports. This written report is an example of a cumulative written report which is a study that includes results of several different tests run on different days.
- Test study appointment. This is the day the results were generated and reported to the ordering physician or to the responsible person. Tests may exist run on a particular patient'southward samples on different dates. Since a patient may accept multiple results of the same test from different days, it is important that the report includes this information for correct estimation of results.
- Name of dr. or legally authorized person ordering the test(s). This information enables the lab to forward your results to the person who requested the test(south). Sometimes a report will too include the proper noun of other health practitioners requesting a copy of your report. For example, a specialist may order tests and request that a copy of the results be sent to your primary healthcare provider.
Other elements establish on reports bargain with the specimen that was collected and with the test itself:
- Specimen source, when appropriate. Some tests can be performed on more than 1 type of sample. For example, protein can exist measured in blood, urine orcerebrospinal fluid, and the results from these different types of specimens can indicate very different things.
- Date and time of specimen drove. Some test results may be affected by the day and time of sample collection. This information may help your health practitioner interpret the results. For example, blood levels of drugs are affected past the fourth dimension a dose of the drug was last taken, so results of the test and its interpretation can be affected past when the sample was collected.
- Laboratory accretion number. Number(s) assigned to the sample(southward) when it arrives at the laboratory. Some labs will have a unmarried accession number for all your tests and other labs may have multiple accession numbers that help the lab identify the samples.
- Name of the test performed. Examination names are often abbreviated on lab reports. You may want to expect for abbreviated test names in the pull down bill of fare on the home page of this site or type the acronym into the search box to notice data on specific tests.
- Test effect. Some results are written as numbers when a substance is measured in a sample equally with a cholesterol level (quantitative). Other reports may simply requite a positive or negative consequence every bit in pregnancy tests (qualitative). Notwithstanding others may include text, such every bit the name of bacteria for the result of a sample taken from an infected site.
- Abnormal test results. Lab reports volition often draw attention to results that are abnormal or outside the reference range (run across "Reference intervals" below) by setting them apart or highlighting them in some manner. For case, "H" adjacent to a result may mean that it is higher than the reference range. "Fifty" may mean "low" and "WNL" usually means "within normal limits."
- Critical results. Those results that are dangerously abnormal must be reported immediately to the responsible person, such as the ordering dr.. The laboratory will often draw attending to such results with an asterisk (*) or something similar and will normally note on the report the engagement and fourth dimension the responsible person was notified.
- Units of measurement (for quantitative results). The units of measurement that labs employ to report your results can vary from lab to lab. Information technology is similar to the way, for example, your health practitioner chooses to record your weight during an exam. He or she may decide to note your weight in pounds or in kilograms. In this same style, labs may choose to use different units of measurement for your exam results. Regardless of the units that the lab uses, your results will be interpreted in relation to the reference ranges supplied by the laboratory.
- Reference intervals (or reference ranges). These are the ranges in which "normal" values are expected to fall. The ranges that appear on your report are established and supplied by the laboratory that performed your exam. They are made bachelor to the health practitioner who requested the test(s) and to other health intendance providers to assist in the interpretation of the results. For more on this, see the commodity on Reference Ranges and What They Mean.
- Interpretation of results. In sure circumstances, the lab may note on the report what certain exam results may indicate.
- Condition of specimen. Any pertinent information regarding the condition of specimens that practice not meet the laboratory'south criteria for acceptability will be noted. This type of data may include a variety of situations in which the specimen was not the all-time possible sample needed for testing. For case, if the specimen was not collected or stored in optimal conditions or if it was visually apparent that a blood sample washemolyzed orlipemic, it will be noted on the report. In some cases, the condition of the specimen may preclude analysis (the test is not run and results are not generated) or may generate additional comments directing the use of circumspection in interpreting results.
- Deviations from test preparation procedures. Some tests have specific procedures to follow before a sample is nerveless or a examination is performed. If such procedures are non followed for some reason, it may be noted on the written report. For instance, if a patient forgets to fast before having a glucose test performed, the study may reflect this fact.
- Medications, health supplements, etc. taken by the patient. Some tests results are afflicted by medications, vitamins and other health supplements, so laboratories may obtain this data from the exam request course and transcribe information technology onto the lab report.
Become the latest lab testing news & updates from our bi-monthly newsletter
Your privacy is important to usa
Source: https://www.testing.com/articles/how-to-read-your-laboratory-report/
Post a Comment for "How to Read a Blood Panel Report"